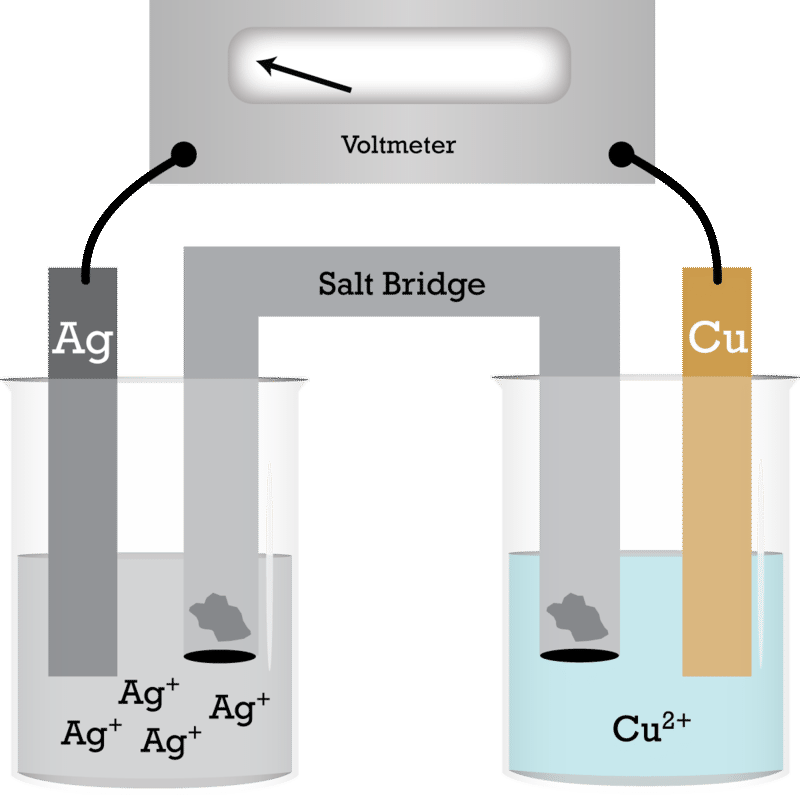
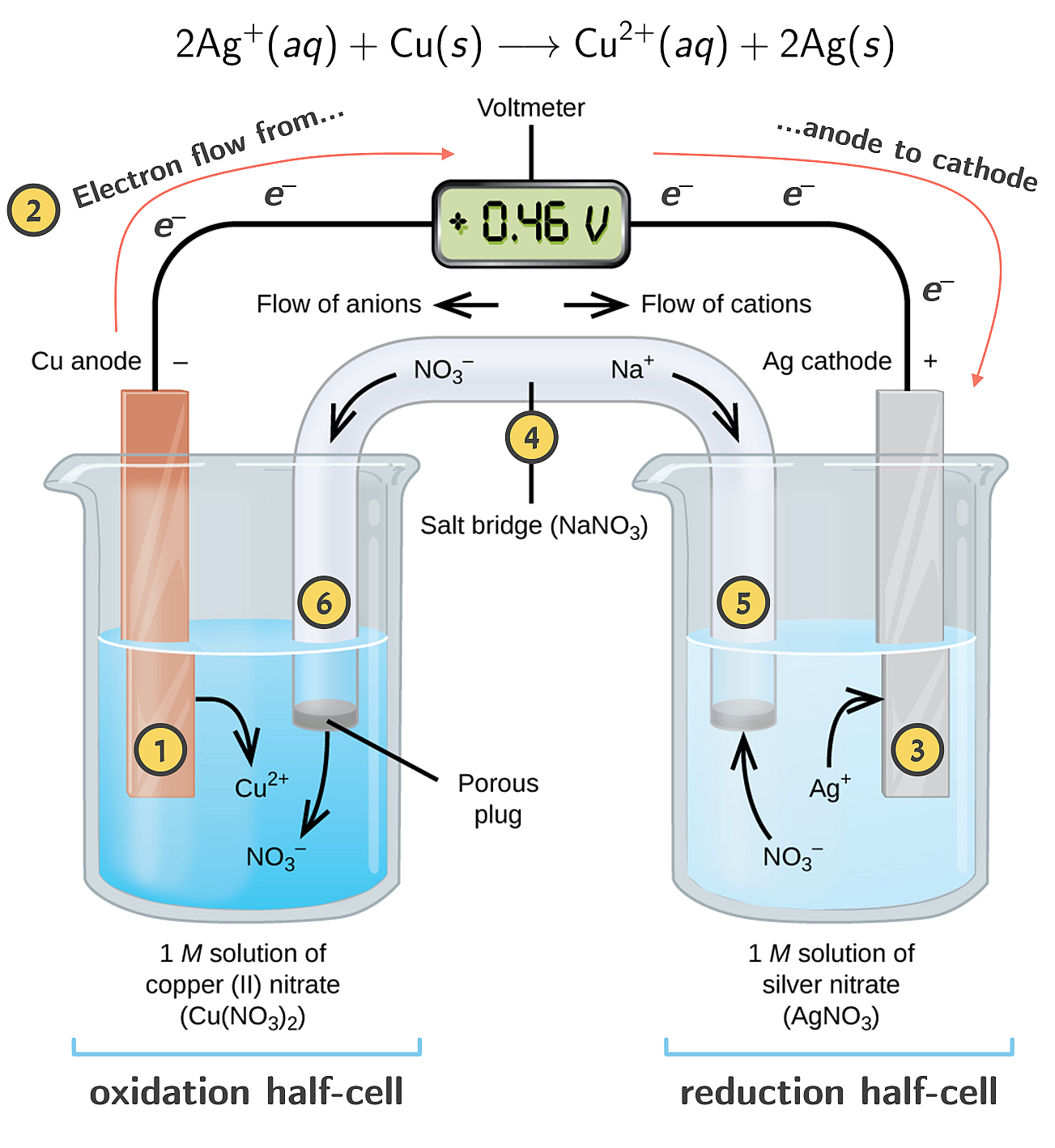
A copper- silver cell is set up. The copper ion concentration in it is 0.10 M. The concentration of silver ion is not known. The cell potential measured 0.422 V. Determine the

a) Write the anode and cathode reactions and the overall reaction occurring in a lead storage battery. (b) A copper-silver cell is up. The copper ion concentration is 0.10 M. The silver

Solve this: 8 A Copper-Silver cell is set up The copper ion - Chemistry - Electrochemistry - 12485096 | Meritnation.com
A copper-silver cell is set up. The copper ion concentration in it is 0.10 M. The concentration of silver ion is not known. - Sarthaks eConnect | Largest Online Education Community

A copper - silver cell is set up. The copper ion concentrations is 0.10 M. The concentration of... - YouTube
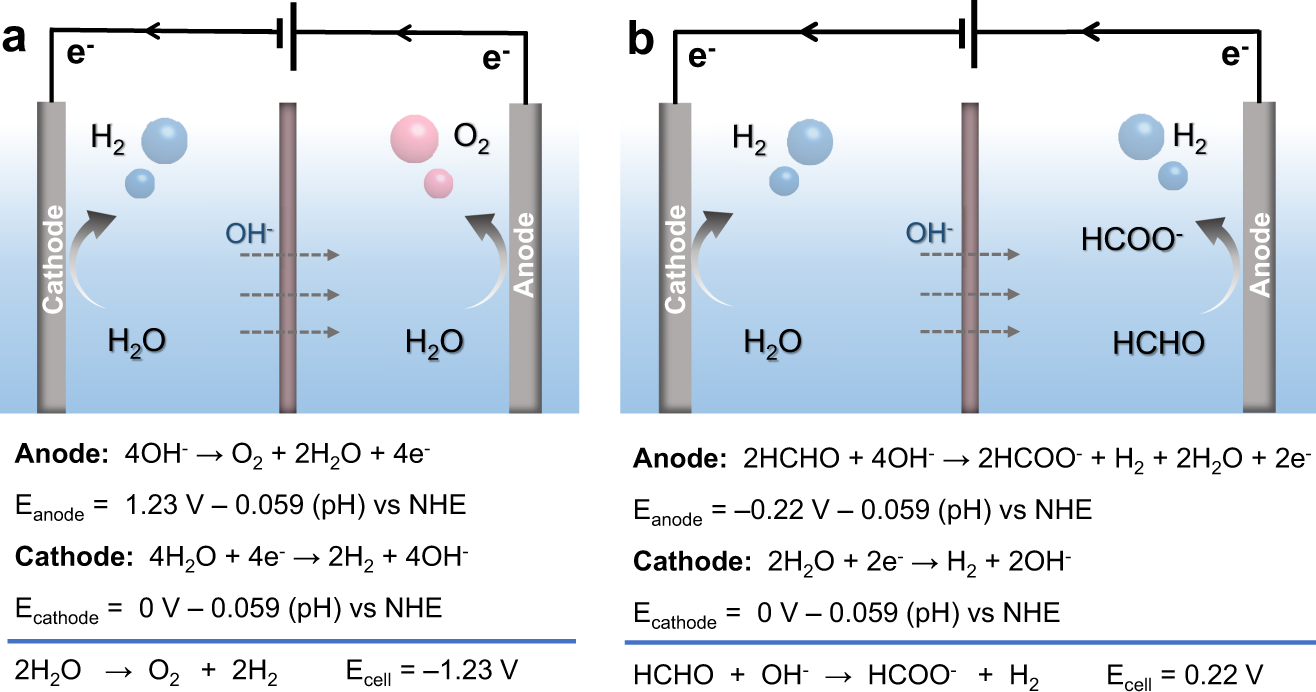
Dual hydrogen production from electrocatalytic water reduction coupled with formaldehyde oxidation via a copper-silver electrocatalyst | Nature Communications

A copper - silver cell is set up. The copper ion concentrations is 0.10 M. The concentration of... - YouTube

A copper-silver cell is set up. The copper ion concentration in it is 0.10 M. The concentration of silver - Brainly.in

A copper - silver cell is set up. The copper ion concentrations is 0.10 M. The concentration of... - YouTube

13.A copper-silver cell is up. The copperion concentration is 0.10M. The concentration of Silver ions is not known. The cell potential was found to be 0.422V.Determine the concentration Silver ion in
A copper-silver cell is set up. The copper ion concentration is 0.10 M. The Ecell = 0.422 V. Determine the concentration of [Ag^+] in the cell if - Sarthaks eConnect | Largest Online Education Community

13.A copper-silver cell is up. The copperion concentration is 0.10M. The concentration of Silver ions is not known. The cell potential was found to be 0.422V.Determine the concentration Silver ion in